 Group 18 on extreme right side position contains noble gases ( He, Ne, Ar, Kr, Xe and Rn ).In the transition elements, valence electrons are present in more than one shell. Elements present in groups 3 to 12 in the middle of the periodic table are called transition elements.The alkaline earth metals are metallic elements found in the group 2 of the periodic table.Group 1 contains alkali metals (Li, Na, K, Rb, Cs and Fr).Periods - In periodic table elements are arranged in a series of rows. Elements of the same period have the same number of electron shells.Each column is called a group. All elements in a group have similar chemical and physical properties because they have the same number of valence electrons. Groups - There are 18 vertical columns in the periodic table.The Modern periodic law states that The chemical and physical properties of elements are a periodic function of their atomic numbers. Modern periodic table is based on the modern periodic law. Periodic Table: It is the table of chemical elements arranged in order of atomic number such that elements with similar atomic structure appear in the vertical columns. Position of Hydrogen could not be assigned in a periodic table.ī.Structure of the modern periodic table:.Wrong order of atomic masses of some elements could not be explained.The position of isotopes could not explained.Mendeleev's periodic table could accomodate noble gases when they were discovered.Mendeleev's periodic table could predict the properties of several elements on the basis of their position in the periodic table.These elements discovered later and named as Gallium, Germanium and Scandium.Mendeleev not only predicted the existence of Eka-Aluminium, Eka-silicon and Eka-Boron but also described the general physical properties of these elements.Mendeleev also left gaps in his periodic table for undiscovered elements like aluminum, silicon and Boron in his periodic table and named them Eka-Aluminium, Eka-silicon and Eka-Boron.The elements with similar properties comes into same group.Mendeleev’s Periodic Table contains seven horizontal rows and nine vertical columns. The horizontal rows are called as periods and vertical columns are called groups. Mendeleev classified periodic table into horizontal rows and vertical coloumns.Mendeleev classified elements according to their atomic masses and arranged these elements in increasing order of their atomic masses. 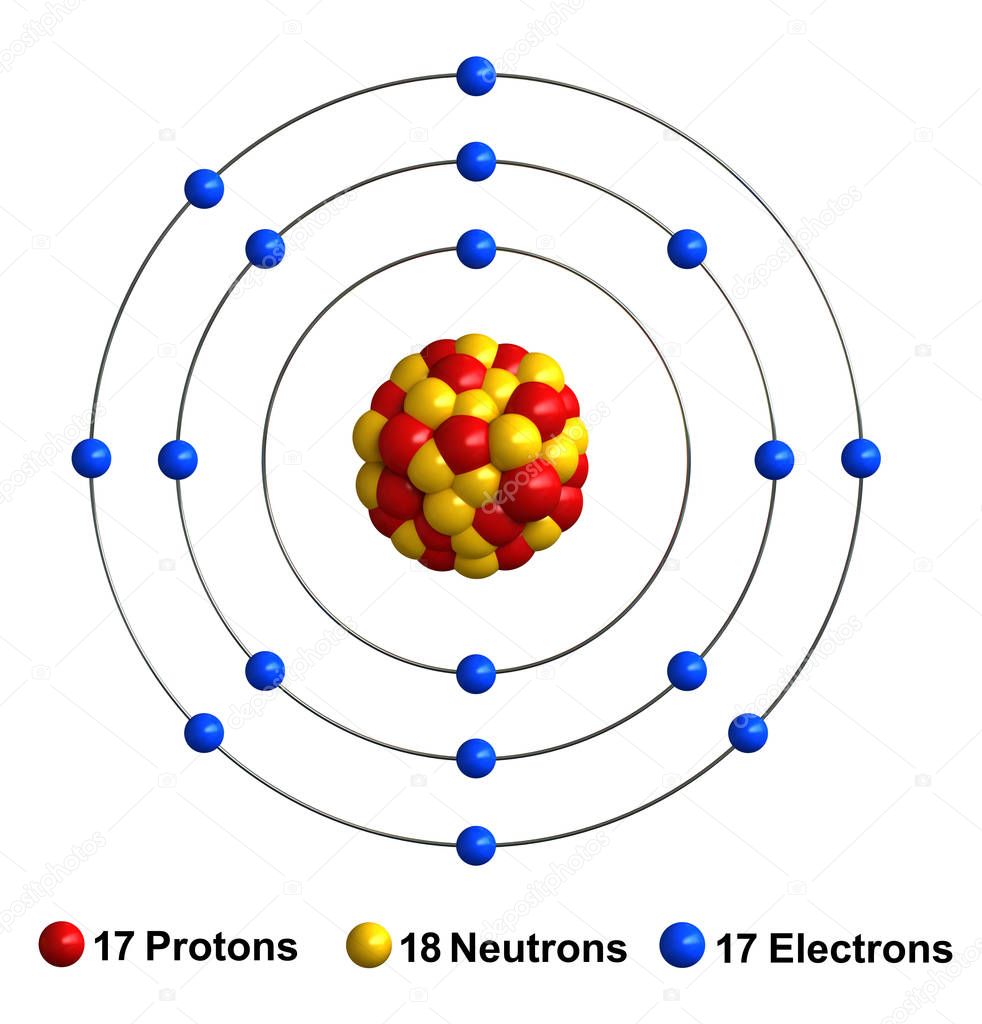
According to Mendeleev’s Periodic Law, “ Physical and chemical properties of elements are periodic function of their atomic masses”.This happens as the tendency of an atom to gain electrons increases due to increase in nuclear charge, the valence electrons are pulled in strongly by the nucleus and it becomes easier for an atom to gain electrons. This happens as the tendency of an atom to lose electrons decreases due to gradual increase in the number of protons and nuclear charge.ĩF has the highest nonmetallic character because non-metallic character of an elements increases as we move from left to right in a period of the periodic table. 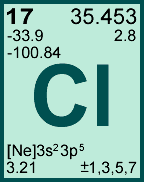
Due to large positive charge on the nucleus, the electrons are pulled closer to the nucleus and the size of atom decreases.ġ1Na has the highest metallic character because metallic character of an elements decreases as we move from left to right in a modern periodic table. According to the trend, as we move from left to right atomic size of an atoms decreases. Due to large positive charge on the nucleus, the electrons are pulled closer to the nucleus and the size of atom decreases.ĤBe has smallest atomic radius because 19K, 3Li, 11Na are present in same group 1 but Be is present in group 2. The atomic number of elements increases which means the number of protons and electrons in the atoms increases. This happens as the tendency of an atom to lose electrons increases due to decrease in nuclear charge and increase in numbers of shell.ġ1Na has largest size among these because according to the trend, atomic radius decreases as we move from left to right in a period of the periodic table. According to the trend, electropositive character of an elements increases as we move from top to bottom in a group of the periodic table. 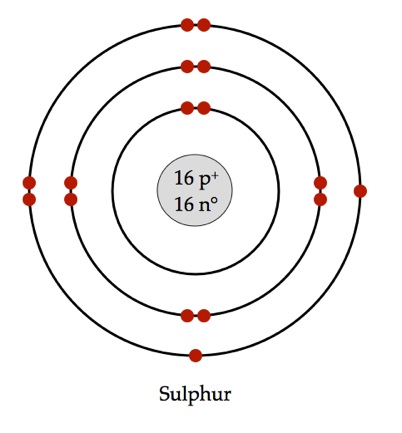
K,L,M.Ģ0Ca, 4Be belong to second group because these elements have 2 electrons in its outermost shell.ĨO is the most electronegative element among these because electronegativity increases as we move from left to right in a period of the periodic table.ġ3Al is the most electropositive element among these because 4Be, 6C, 8O, 5B belong to same period, but 13Al belong to next period. Electronic configuration of the following elements is:ġ4He, 11Na, 15P belong to the third period because according to their electronic configuration, each element contains three shell i.e. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
